A large number of people—including many physicians—believe that obesity is a consequence of a person’s moral character or poor choices. The reality could not be farther from the truth. Obesity is a chronic, relapsing neurobehavioral disease driven by the collision of ancient biology with a modern food environment.1 Here’s the science behind why.
The Obesity Epidemic Is New—Our Biology Is Not
There are many contributors to obesity, both internal and external. The Obesity Society has catalogued dozens of potential contributors spanning biological, psychological, social, economic, and environmental categories.2
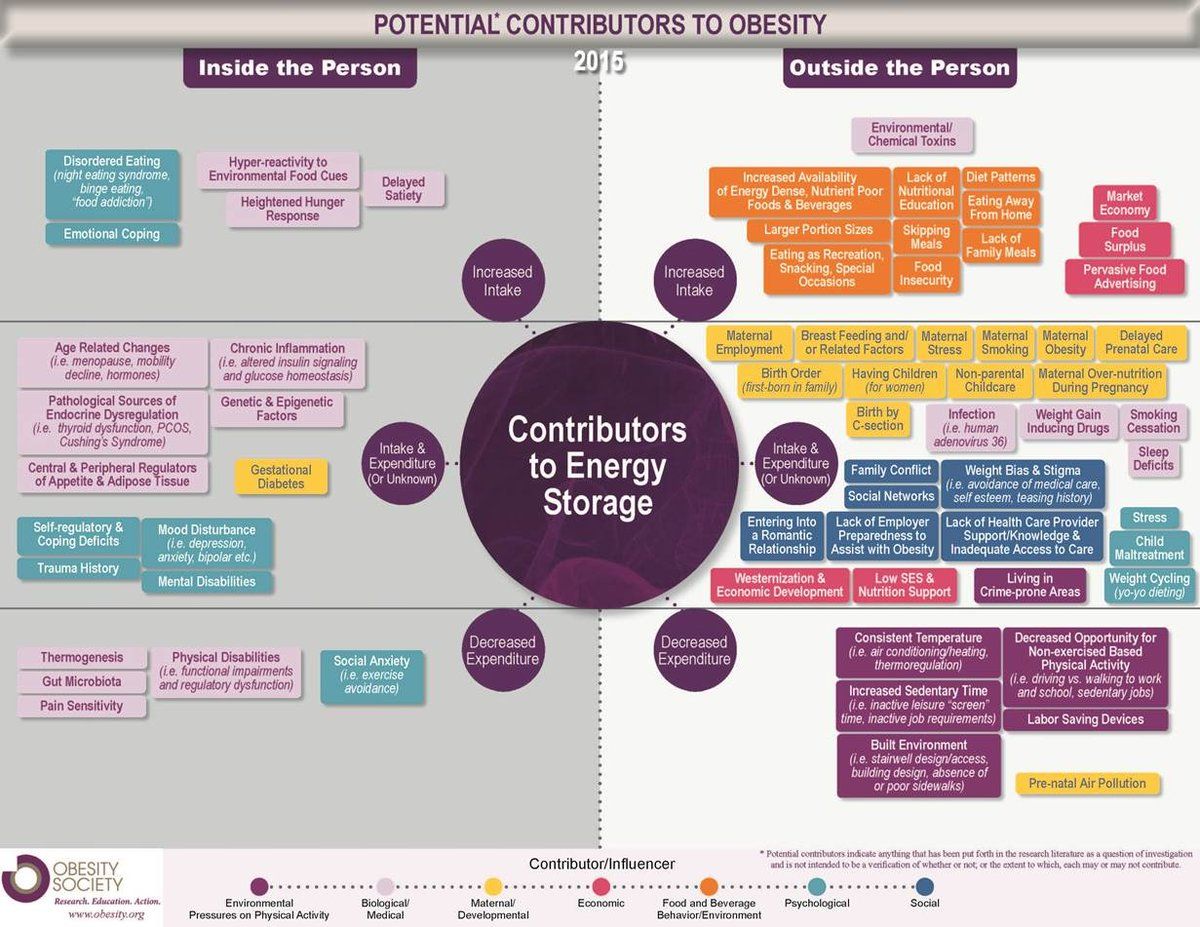
Figure 1. Potential contributors to obesity, spanning factors both inside and outside the person. Source: The Obesity Society, 2015.
But the obesity epidemic itself is a relatively recent phenomenon, only emerging since the 1970s. Most researchers believe it stems from the convergence of our deep evolutionary biology with the modern food environment.3
The numbers paint a stark picture. Calorie intake over the last 50 years has steadily increased alongside the prevalence of obesity throughout the United States. Our best calculations estimate that the modern obesity epidemic is the result of an additional 200 calories of food consumed per day—a modest surplus that, compounded over time, leads to significant weight gain.4
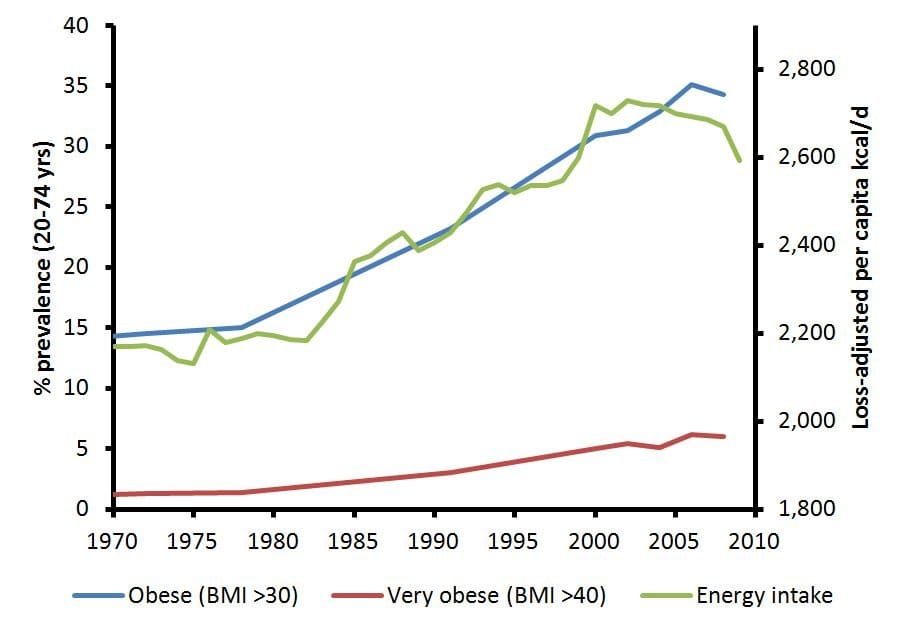
Figure 2. US prevalence of obesity (BMI >30) and severe obesity (BMI >40) from 1970–2010, superimposed with average daily calorie consumption. Adapted from Dhurandhar, 2015.4
The food environment has also become economically skewed. Over the past several decades, the price of fresh fruits and vegetables has risen dramatically compared to sugars, fats, and carbonated drinks, making energy-dense, nutrient-poor foods the more affordable and convenient option for many families.
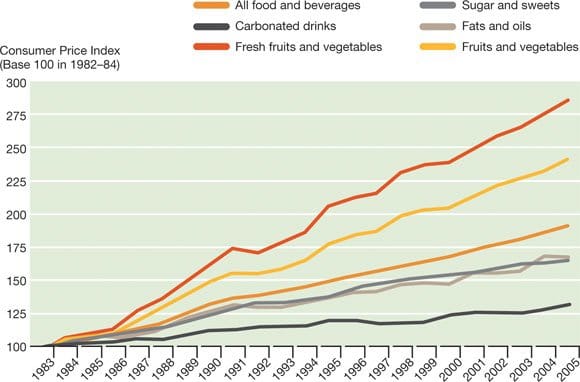
Figure 3. Consumer Price Index for various food categories (1983–2005). Fresh fruits and vegetables have risen in price far faster than processed foods and sweetened beverages.
So if we’re eating more, why don’t we just stop?
Many have tried. People have been dieting on and off their whole lives, and losing weight—let alone maintaining that loss—is extraordinarily difficult. Research shows that most of the weight lost through dieting is regained within a few years. By five years out, more than 80% of lost weight has typically returned.5
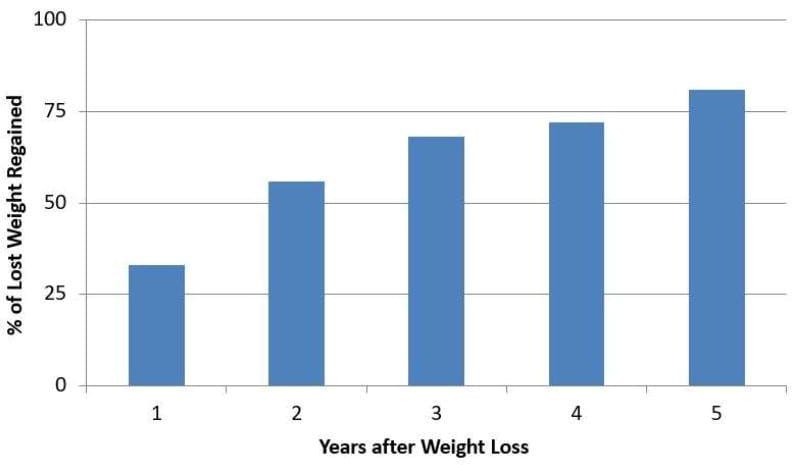
Figure 4. Percentage of lost weight regained over five years following weight-loss intervention. Anderson et al., 2001.
Maybe people just aren’t dieting “correctly”? Maybe they’re eating too many whole grains and fruit (this one always gets a laugh). But to truly understand why people struggle to lose weight, we need to look deeper—at the biological feedback systems that evolved over millions of years to conserve energy.
The Homeostatic System: Your Body’s Built-In Defense Against Weight Loss
The first major player is the homeostatic system. Centered primarily in the hypothalamus, with nerve inputs from many parts of the brain, this system regulates energy expenditure and hunger-satiety signaling. It has developed over millions of years to do one thing exceptionally well: defend against weight loss.6
To understand why, consider our ancestors. They subsisted as hunter-gatherers, and foraging for food was difficult because it was not abundant. During times of shortage, neural systems evolved to increase hunger, decrease energy expenditure, and amplify motivation and pleasure from energy-dense foods.7 This was a survival advantage—our ancestors would have died of starvation without systems to promote the seeking and storage of energy.

Figure 5. Our ancestors subsisted as hunter-gatherers in environments where food was scarce and unreliable, selecting for neural systems that promoted energy conservation.
The problem? The modern food environment bears no resemblance to the one these systems were designed for. Today we have easy, 24/7 access to hyperpalatable, highly rewarding, energy-dense foods—sugar, salt, and fat-added, ultra-portioned, ultra-available, delivered right to your front door.8
In 1994, researchers identified a polypeptide secreted by fat cells in amounts proportional to total body fatness. This adipokine, known as leptin, turned out to be integral in controlling the homeostatic system. When someone loses weight, leptin levels drop, triggering activation of hunger centers and a decrease in energy expenditure. Leptin essentially functions as a thermostat for body weight: if fat mass decreases, leptin decreases, and the brain increases hunger while decreasing energy expenditure in an effort to regain weight around a non-rigid set point.

Figure 6. The leptin feedback loop. Fat tissue secretes leptin, which signals the hypothalamus to regulate food intake and energy expenditure. When fat mass decreases, reduced leptin triggers increased hunger and decreased energy expenditure.
Unfortunately, this system does not appear to be nearly as robust against weight gain—likely because it evolved during a time of food scarcity when gaining too much weight simply wasn’t the threat that starvation was.
The magnitude of these compensatory changes is striking. For every kilogram of lost weight, calorie expenditure decreases by about 20–30 kcal per day, while appetite increases by about 100 kcal per day above the baseline level prior to weight loss.6
In summary, the homeostatic system defends against changes in body weight and will increasingly alter physiology to return body weight within a set range, making it profoundly difficult to sustain weight loss through willpower alone.
The Hedonic System: When Food Hijacks Your Reward Circuits
The second major player is the hedonic system. This is a complex neural circuit with inputs from all over the brain, essential for reinforcement and motivating goal-directed behavior.9

Figure 7. Brain regions involved in energy balance regulation, including the prefrontal cortex (PFC), nucleus accumbens (NAc), amygdala (AMY), arcuate nucleus (ARC), and lateral hypothalamus (LH), among others.
Over time, external food cues (advertising), food sensory information (taste and smell), and concentrated ingredients become learned stimuli, conditioned by increases in dopamine to drive eating. Crucially, most of these processes occur below conscious control.10
Using functional MRI, researchers have shown that people with obesity have increased activity in brain regions that drive anticipatory feeding but decreased activity in reward-related regions. In other words, obesity drives the wanting but not the contentment, which can lead to overeating, as ever-greater stimulation is needed to achieve satisfaction.
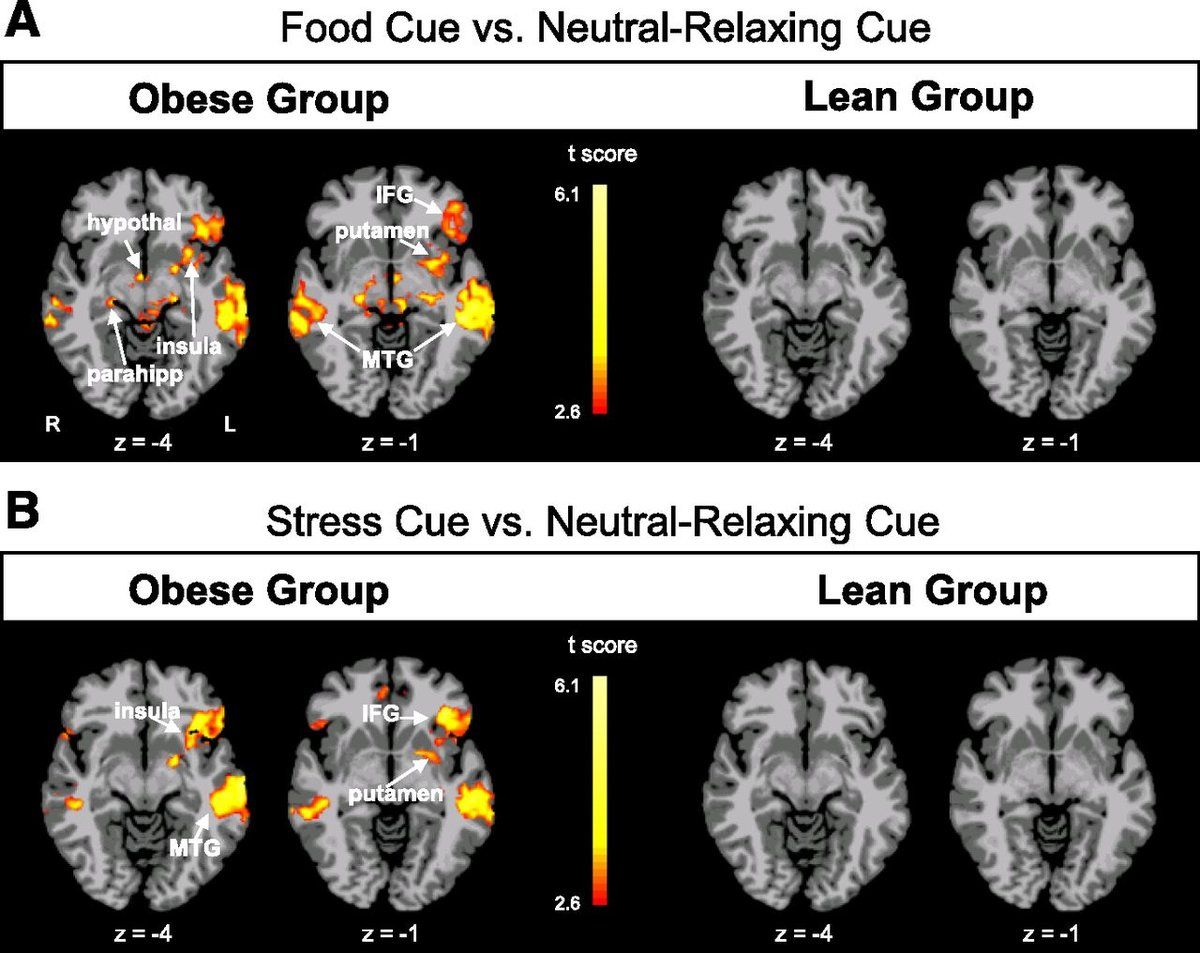
Figure 8. fMRI scans comparing brain activation in obese vs. lean individuals in response to food cues (top) and stress cues (bottom). The obese group shows markedly greater activation in the hypothalamus, insula, putamen, and other regions involved in craving and reward.
The brain scans are remarkably similar to patterns seen in substance use disorders, with comparable changes in dopamine D2 receptor availability and orbitofrontal cortex metabolism between obesity and cocaine use disorder.10
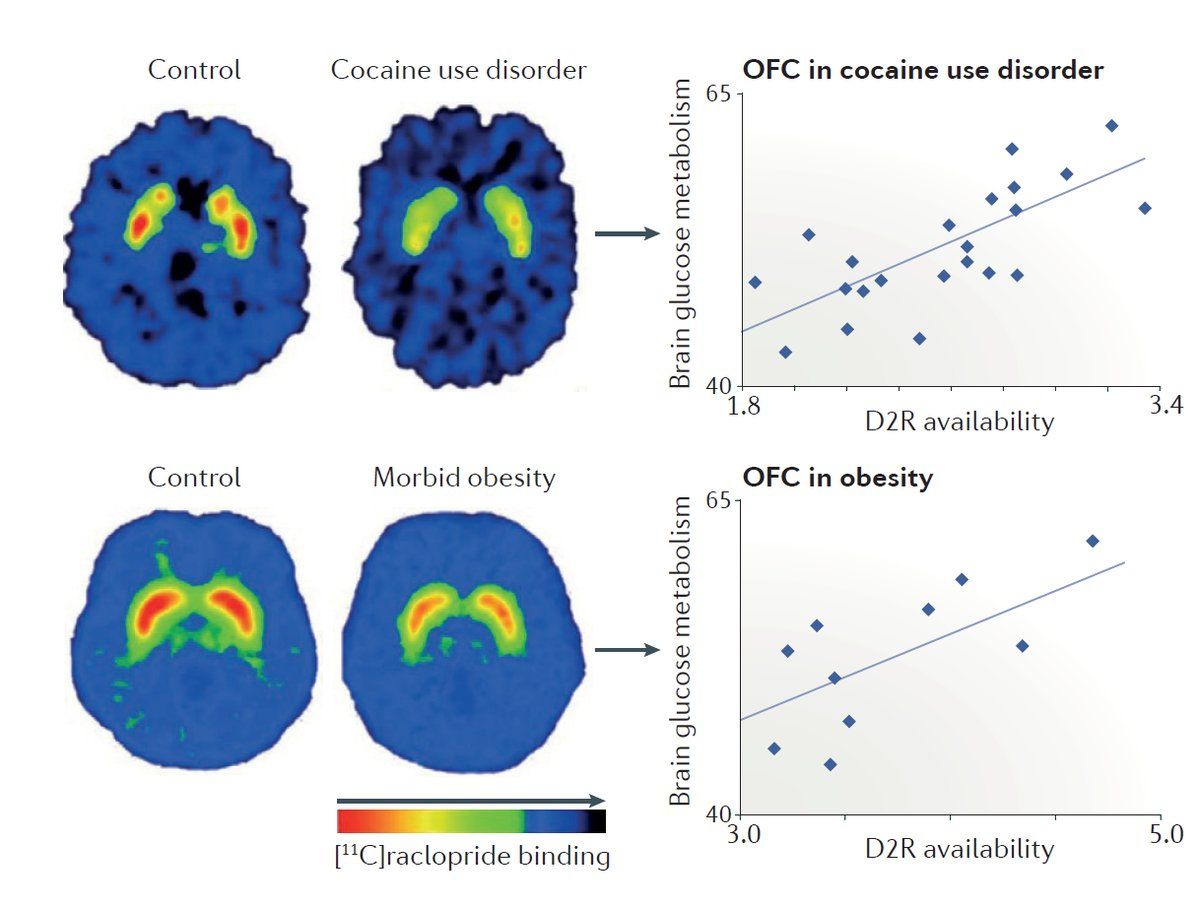
Figure 9. PET scan comparisons showing reduced dopamine D2 receptor (D2R) availability in both cocaine use disorder (top) and obesity (bottom), with similar positive correlations to orbitofrontal cortex (OFC) metabolism.
The Executive System: Willpower Has Real Limits
But some people do lose weight and keep it off, right? That’s true—and those successful restrained eaters engage the third area of this puzzle: the executive system, centered in the prefrontal cortex (PFC).
This area of the brain is mostly under conscious control and works to inhibit hedonic signaling. It’s involved in self-regulation and planning, and successful restrained eaters can engage the PFC to emphasize delayed rewards and selective attention. However, obesity itself can blunt prefrontal cortex activity, making conscious override harder to achieve.
And even in people who successfully engage executive control, research shows that acute stressors can easily disinhibit these circuits, decoupling PFC control and leading to overeating. From an evolutionary perspective, a hedonic inhibitory system may have developed under ecological and social pressures—an individual needed to store food for times of shortage, share with a group to cultivate relationships, and care for offspring. But in a world of unlimited food availability, these brakes are woefully insufficient.
Putting It All Together

Figure 10. Simplified schematic of the interplay between neural inputs, brain areas (prefrontal cortex, mesolimbic dopamine system, hypothalamus), motivational processes (inhibitory control, hedonic hunger, homeostatic hunger), and behavioral outputs (feeding behavior, delay discounting, selective attention).
These three microsystems—homeostatic, hedonic, and executive—interact with one another and influence each other’s activity. They are driven by millions of biological factors whose functions are determined by our genetics and epigenetics, which likely explain the wide variation in obesity susceptibility and weight-loss success between individuals.
Twin studies estimate that genetics accounts for roughly 40–70% of the variability in adult BMI, and genome-wide association studies show that most of the genetic risk for weight gain is encoded in genes involved in central nervous system development.10,11 As Dr. Francis Collins, former Director of the NIH, once said: “Genes load the gun, but the environment pulls the trigger.”
Even in the most ideal circumstances, losing weight is hard because of biology that is maladapted for the modern food environment. Add external influences like job and food insecurity, lack of access to healthy food, endocrine disruptors, environmental toxins, and pervasive weight stigma, and it becomes increasingly difficult.12,13
Why This Matters
This is, admittedly, a simplified account of what is an extraordinarily complex neuroendocrine system that science is still working to fully elucidate. But the evidence makes a powerful case: obesity occurs through biological mechanisms that operate largely outside of conscious control, and it is far more than a simple choice.
Understanding this science isn’t just an academic exercise. Weight discrimination is comparable in prevalence to racial discrimination, particularly among women.12 Healthcare workers demonstrate implicit and explicit biases toward people with obesity,13 leading to delays and avoidance in care.14 And the common framing of obesity as a moral failure causes real harm to real people.
Obesity is a chronic, relapsing neurobehavioral disease. The people living with it deserve our compassion, our understanding, and access to evidence-based treatments—not our judgment.
References
Lazarus. “What is Obesity?” Obesity Medicine Association.
Kopelman P. Symposium 1: Overnutrition: Consequences and solutions. Foresight Report: The obesity challenge ahead. Proc Nutr Soc. 2009;69:80–85.
Swinburn B, Egger G, Raza F. Dissecting obesogenic environments: the development and application of a framework for identifying and prioritizing environmental interventions for obesity. Prev Med. 1999;29(6):563–570.
Dhurandhar N. TOS opening session. Presented at ObesityWeek; Nov. 2–6, 2015; Los Angeles.
Montesi L, et al. Long-term weight loss maintenance for obesity: a multidisciplinary approach. Diabetes Metab Syndr Obes. 2016;9:37.
Polidori D, et al. How strongly does appetite counter weight loss? Quantification of the feedback control of human energy intake. Obesity. 2016;24(11):2289–2295.
Grannell A, et al. Obesity and responsibility: Is it time to rethink agency? Obes Rev. 2021:e13270.
Llewellyn C, Wardle J. Behavioral susceptibility to obesity: gene–environment interplay in the development of weight. Physiol Behav. 2015;152:494–501.
Morton GJ, Meek TH, Schwartz MW. Neurobiology of food intake in health and disease. Nat Rev Neurosci. 2014;15(6):367–378.
Stice E, Burger K. Neural vulnerability factors for obesity. Clin Psychol Rev. 2019;68:38–53.
Elks CE, et al. Variability in the heritability of body mass index: a systematic review and meta-regression. Front Endocrinol. 2012;3:29.
Puhl RM, et al. Perceptions of weight discrimination: prevalence and comparison to race and gender discrimination in America. Int J Obes. 2008;32(6):992–1000.
Lawrence BJ, et al. Weight bias among health care professionals: a systematic review and meta-analysis. Obesity. 2021.
Phelan SM, et al. A model of weight-based stigma in health care and utilization outcomes: evidence from the Learning Health Systems Network. Obes Sci Pract.
